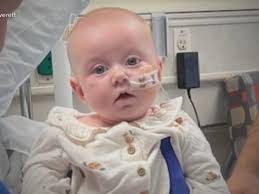
A Kentucky mother is sharing her fears after her 4-month-old daughter was hospitalized with infant botulism, reportedly after consuming a recently recalled infant formula brand.
“You just feel helpless,” Hanna Everett told ABC News, explaining that her baby, Piper, became ill after ingesting ByHeart Whole Nutrition Infant Formula.
From Aug. 1 through Nov. 10, 84 infants across the U.S. received treatment for infant botulism, according to an FDA news release. Thirty-six of those cases involved powdered infant formula, and 15 of those babies had consumed ByHeart Whole Nutrition Infant Formula.
On Saturday, Nov. 8, preliminary lab results showed bacteria capable of producing botulinum toxin in a can of formula used by a sick infant. The FDA subsequently requested a recall of two ByHeart formula batches. By Tuesday, Nov. 11, the company voluntarily expanded the recall nationwide to include all its formula products, both canned and single-serve.
Botulism is a rare but serious illness caused by toxins produced by Clostridium botulinum and related bacteria, per the CDC. These toxins can attack the nervous system and lead to difficulty breathing, paralysis, or even death. Early warning signs in infants may include constipation, poor feeding, difficulty swallowing, reduced facial expression, and a loss of head control.
All 15 babies linked to the brand’s formula required hospitalization, but the agency noted that no deaths had been reported.
That same Saturday, Hanna noticed Piper was no longer eating or sleeping as usual, according to ABC News and WKYT. “Thirty minutes later, I had a friend send me that recall, and I was like, ‘Oh my god,’ ” she said. Piper has remained in the hospital but is showing major improvement.
“She’s gonna make a full recovery,” Hanna told WKYT. “They said we were very lucky to catch it as early as we did.”
ByHeart released a statement on its website acknowledging the expanded recall as a precaution to protect infants, and said that neither the company nor federal agencies had found botulinum spores or toxins in unopened cans of formula.
“We worked with a third-party accredited laboratory to conduct testing on our product,” Dr. Devon Kuehn, ByHeart’s chief medical officer, told ABC News. The company also emphasized transparency with the FDA and CDC throughout inspections and testing.
:max_bytes(150000):strip_icc():focal(749x0:751x2):format(webp)/Baby-formula-recalled-over-botulism-concerns-111325-ce205dfbaf294cd59298c2513344254a.jpg)
“Our number one priority is protecting babies,” said Mia Funt, co-founder and president of ByHeart, in a statement. “We express our deepest sympathy to our ByHeart families and all those impacted. Our focus is ensuring families are aware of the recall and understand what steps to take.”
The CDC advises parents and caregivers to avoid using any ByHeart Whole Nutrition infant formula and to thoroughly wash surfaces or feeding items that may have come into contact with it. If an infant shows symptoms of botulism, immediate medical care is crucial.
For baby Piper, swift recognition made a life-changing difference, ABC News reported.
On Tuesday, Nov. 11, Hanna posted online about her commitment to raising awareness:
“We will continue to advocate and fight for our daughter! Something as important as feeding your child should never come with concerns of their safety!”